
Alpha decay occurs in such nuclei as a means of increasing stability by reducing size. A nucleus with 210 or more nucleons is so large that the strong nuclear force holding it together can just barely counterbalance the electromagnetic repulsion between the protons it contains. The strength of the attractive nuclear force keeping a nucleus together is thus proportional to the number of the nucleons, but the total disruptive electromagnetic force of proton-proton repulsion trying to break the nucleus apart is roughly proportional to the square of its atomic number. 
However, the nuclear force is also short-range, dropping quickly in strength beyond about 1 femtometer, while the electromagnetic force has an unlimited range. The nuclear force holding an atomic nucleus together is very strong, in general much stronger than the repulsive electromagnetic forces between the protons. Gamow solved a model potential for the nucleus and derived, from first principles, a relationship between the half-life of the decay, and the energy of the emission, which had been previously discovered empirically and was known as the Geiger–Nuttall law. Classically, it is forbidden to escape, but according to the (then) newly discovered principles of quantum mechanics, it has a tiny (but non-zero) probability of " tunneling" through the barrier and appearing on the other side to escape the nucleus. The alpha particle is trapped inside the nucleus by an attractive nuclear potential wellĪnd a repulsive electromagnetic potential barrier. See also: Alpha particle § History of discovery and useĪlpha particles were first described in the investigations of radioactivity by Ernest Rutherford in 1899, and by 1907 they were identified as He 2+ ions.īy 1928, George Gamow had solved the theory of alpha decay via tunneling. The helium is brought to the surface as a by-product of natural gas production. Because of their relatively large mass, the electric charge of +2 e and relatively low velocity, alpha particles are very likely to interact with other atoms and lose their energy, and their forward motion can be stopped by a few centimeters of air.Īpproximately 99% of the helium produced on Earth is the result of the alpha decay of underground deposits of minerals containing uranium or thorium. There is surprisingly small variation around this energy, due to the strong dependence of the half-life of this process on the energy produced. Unlike beta decay, it is governed by the interplay between both the strong nuclear force and the electromagnetic force.Īlpha particles have a typical kinetic energy of 5 MeV (or ≈ 0.13% of their total energy, 110 TJ/kg) and have a speed of about 15,000,000 m/s, or 5% of the speed of light. Like other cluster decays, alpha decay is fundamentally a quantum tunneling process. It is the most common form because of the combined extremely high nuclear binding energy and relatively small mass of the alpha particle. Exceptionally, however, beryllium-8 decays to two alpha particles.Īlpha decay is by far the most common form of cluster decay, where the parent atom ejects a defined daughter collection of nucleons, leaving another defined product behind. 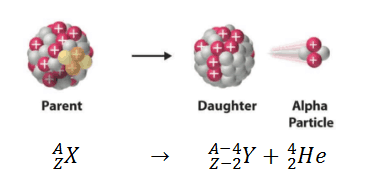
In practice, this mode of decay has only been observed in nuclides considerably heavier than nickel, with the lightest known alpha emitters being the lightest isotopes (mass numbers 104–109) of tellurium (element 52). Theoretically, it can occur only in nuclei somewhat heavier than nickel (element 28), where the overall binding energy per nucleon is no longer a maximum and the nuclides are therefore unstable toward spontaneous fission-type processes. While alpha particles have a charge +2 e, this is not usually shown because a nuclear equation describes a nuclear reaction without considering the electrons – a convention that does not imply that the nuclei necessarily occur in neutral atoms.Īlpha decay typically occurs in the heaviest nuclides. For example, uranium-238 decays to form thorium-234. It has a charge of +2 e and a mass of 4 u. An alpha particle is identical to the nucleus of a helium-4 atom, which consists of two protons and two neutrons. Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (helium nucleus) and thereby transforms or 'decays' into a different atomic nucleus, with a mass number that is reduced by four and an atomic number that is reduced by two.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
